While the pandemic progresses, discussions arise on how we might ‘open up’ society again. Assessing the immune response against the virus in those who have been in contact with SARS-CoV-2 would be essential for that. Their immune system has fought off the virus and, therefore, they may be immune. We don’t know yet whether this immunity is indeed protective for re-infection, or that it is long-lasting. Much research is in the working on that, next to clinical trials in which patients with severe symptoms receive plasma from recovered patients. The thought behind that is that someone else’s antibodies may help the body fight the disease.
Many laboratory tests are in development that detect antibodies directed against the virus. Such tests can determine whether there are antibodies, but ideally also tell you the antibody titer. The titer indicates the level of antibodies present in a blood sample. It is defined as the highest dilution, or lowest concentration, of the blood sample at which an assay can still detect antibodies. The more antibody in the blood, the more you can dilute and still detect it, and the higher the titer.
So how can we test for antibodies? Most of the tests in development (look here for a summary by the FDA) use ELISA or lateral flow techniques.
ELISA

An enzyme-linked Immunosorbent assay (ELISA) is a simple, yet very effective tool to look for antibodies. It uses detection antibodies specific for the protein you are looking for – in this case the antibodies against SARS-COV-2 – that have an enzyme linked to it. This enzyme is then used to induce a color reaction. If the protein is present, you will see a color change, if not, no change happens.
How does it work?
Direct ELISA
The direct ELISA is the technique in its simplest form. In a wells plate, you add your sample of analysis – in this case blood containing the potential antibodies (your antigen of interest) – to ‘coat’ the plate with that antigen. Later you add antigen-specific antibodies to it. These antibodies are directly labeled with an enzyme. Once you have given them time to bind, you wash away any unbound antibodies and then add your reagent to obtain a color reaction.
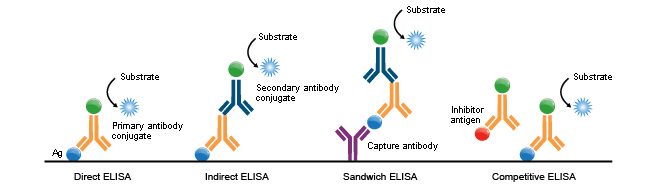
You won’t always have enzyme-labeled specific antibodies to your target antigen available. It would be way too costly for manufacturers to create these for every protein of interest. But that doesn’t mean you cannot do ELISAs with those.
Indirect ELISA
Other techniques that use a bit more complex set up can make use of unlabeled antibodies. Again, in the simplest form, you have the indirect ELISA. This uses the same technique as the direct ELISA, but uses a secondary antibody. So you first add your specific antibody, and after washing away any unbound antibody, you add a secondary antibody directed against the animal species the first antibody was created in. This secondary antibody is enzyme-labeled, allowing for the color reaction to take place.
Sandwich ELISA
You won’t always be able to coat the wells of the test plate with the antigen, in which case you may need to capture it first. That is what a sandwich ELISA does. First, you coat your wells with a capture antibody, specific for your antigen of study, and add your sample to it. After that, you come in again with a specific antibody and a labeled secondary to detect it.
The color intensity in each well can be measured to determine the quantity of the antigen.
Lateral flow assays

Lateral flow assays are also a useful technique for the detection of antibodies. A good example of that technique is the home pregnancy test, which detects the presence of the hormone hCG in urine. The pregnancy test is a little plastic device with an area to apply the urine to and has a screen that gives you one colored line (not pregnant) or two colored lines (pregnant). Similar tests are in development to detect COVID antibodies.
How does that technique work?
The mechanism behind it is very similar to that of ELISA; you detect an analyte of interest by using labeled antibodies specific for the antigen of interest. In the case of testing for SARS-COV-2-specific antibodies, you would take a blood sample and let that run over the test kit. You apply the sample on one end of the pad, called the sample pad. The sample will migrate through the pad to the next section, which contains dried antibodies specific to your analyte (in this case, a general human antibody class such as IgG or IgM). These antibodies have colored particles conjugated to them (often this would be colloidal gold).
After that, the antibody-bound analyte will migrate further into the pad and come across the actual test region (detection region). This is a porous membrane containing one line of specific antibody (detecting the SARS-CoV-2 antibodies) and one line of, again, general IgG or IgM-detecting antibodies. If our antibody of interest was present, it would show a colored line in the test spot, while the control would light up to show you that the test worked.
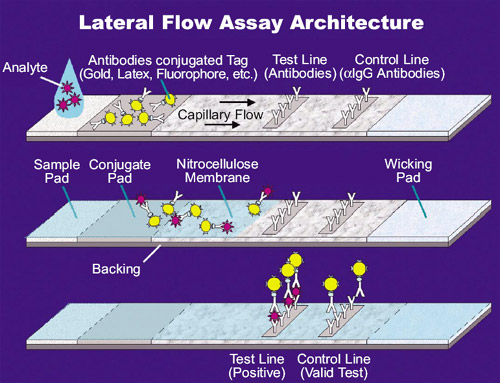
The benefit of this technique is that it doesn’t require special equipment in most cases, and it can be developed into a relatively cheap home test.
The big question: how good are these tests?
As it often is in life in the lab: your data are only as good as the quality of your test. Here, the tests’ techniques are well-established, and we do not need to worry about that (as long as they’re adequately performed by the user). But what we do need to worry about is the specificity of the antibodies we use.
In the ideal situation, they only detect the antibody specific for SARS-CoV-2, and no other viruses. Further, it does catch all of them, so you don’t miss any signal. That’s what we want from a test.
However, often you have antibodies that are not perfect, so they might detect antibodies targeted against other coronaviruses. This is not strange as the viruses are all of the same family and have a lot in common. Or you might have such a perfect detection that you also miss a lot.
Sensitivity and Specificity
The sensitivity and specificity identifies these issues for every test.
Sensitivity defines how well the test can correctly predict whether a sample has the analyte you’re looking for. In other words, how well it can identify true positives.
The specificity, on the other hand, defines how well it can detect a negative sample; the true negatives. We can also calculate the positive and negative predictive value. This indicates the probability that someone who is found positive in the test is truly positive, or someone who is found negative is truly negative.
Now, for testing of COVID-19 antibodies, we, of course, want to ensure anyone who is positive is detected. But it might be even more important to ensure that we do not get false positives. If someone would hear they have immunity, when they actually don’t, that is obviously a problem.
We have to take a close look at these values for all tests in development. An additional issue with the current situation is that still only a relatively small portion of the world population has dealt with this virus, which makes the testing a bit more tricky.
Does having antibodies tell you that you are safe?
The simple answer to that is: we don’t know. We do not know if having antibodies prevents you from getting sick again. If the antibodies do indeed prevent disease, we also don’t know how much of it you need to have. And we don’t know how long this immunity will last.
There are lots of remaining questions. All of this is heavily studied at the moment, but it will take time before we have some clear answers.
What can you do now?
So until then, keep your physical distance to people, wash your hands well and often, and try to stay calm. That’s really the best you can do for now.
If you are struggling with the current situation, please know that it is okay. You are not alone. Check out our previous blogs on online resources for learning and developing skills and side hustles you can do from your home during these COVID-19 times.









